When History Repeats Itself, Patients Pay the Price
This is the moment to break the cycle.
Medicine is filled with moments we look back on and ask, “Why did it take so long?”
History shows repeated periods when the delayed adoption of evidence-based advances contributed to preventable harm. The reasons are often layered: scientific uncertainty, institutional inertia, reimbursement structures, and professional turf battles. Rarely is it as simple as doctors “refusing” to change.
But the outcome is the same.
When proven advances are slowed, patients suffer.
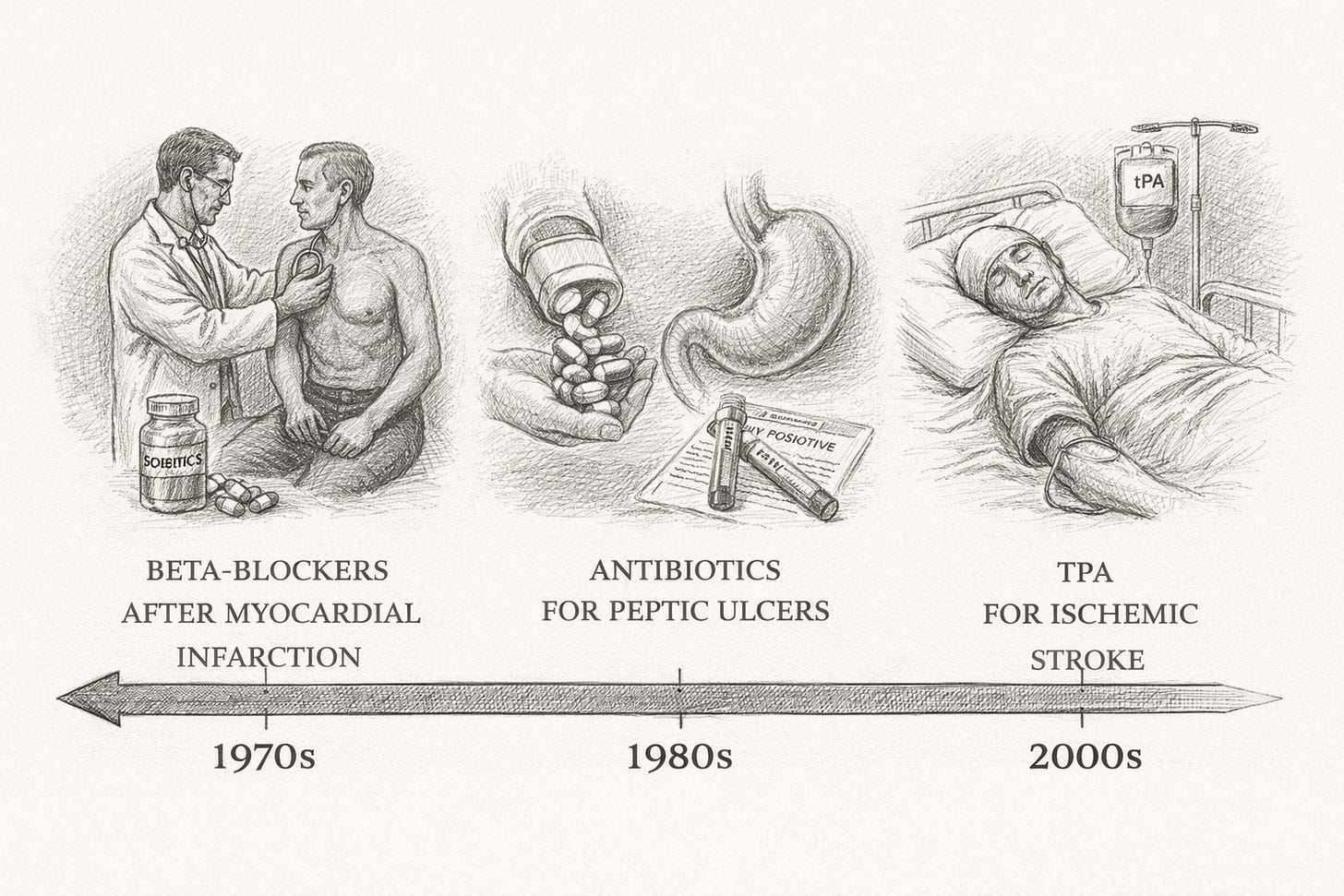
In the 1970s, randomized trials demonstrated that beta-blockers reduced mortality after myocardial infarction. The evidence was strong. Yet routine use lagged for years. Health services research later suggested that delays in adopting beta-blockers and other evidence-based cardiac therapies may have contributed to preventable cardiac deaths before widespread uptake.
In the 1980s, Barry Marshall and Robin Warren demonstrated that most peptic ulcers were caused by Helicobacter pylori and could be treated with antibiotics. The prevailing paradigm — stress and acid — persisted long after evidence said otherwise. Patients endured chronic medication use, repeated hospitalizations, and unnecessary surgeries before antibiotic therapy became standard of care.
In the 1990s, combination antiretroviral therapy dramatically reduced HIV mortality. Yet skepticism, cost barriers, and infrastructure challenges delayed access in many settings. Thousands died waiting for systems to catch up to science.
In the 2000s, tissue plasminogen activator (tPA) was shown to improve outcomes when administered within a defined window after ischemic stroke. Concerns about bleeding risk, logistics, and medicolegal exposure slowed implementation in some regions. Where stroke systems developed slowly, eligible patients missed time-sensitive therapy.
Each decade carries its own version of the same story:
Evidence emerges.
Resistance forms.
Adoption stalls.
Patients pay.
When we look back on the 2020s, dermatology may have its own chapter in this pattern.
Image-Guided Superficial Radiation Therapy (IGSRT) represents a meaningful advancement in the treatment of appropriately selected nonmelanoma skin cancers. For these patients, peer-reviewed data demonstrate excellent local control rates, strong safety outcomes, and favorable cosmetic results. More than 140,000 patients have already been treated with IGSRT over the past eight years.
In the largest patient satisfaction study to date, 99.8% of more than 40,000 patients surveyed reported being satisfied with their decision to undergo GentleCure Image-Guided SRT and would recommend the treatment to others.
Contrast that with survey data from more than 1,000 Mohs patients, in which fewer than 15% reported being both satisfied and willing to recommend their treatment experience.
And yet — despite clinical outcomes, safety data, and overwhelming patient satisfaction — organized opposition has intensified.
The American College of Mohs Surgery (ACMS) has actively advised the AMA and CMS on coding and coverage issues, arguing that image guidance is unnecessary when treating skin cancer with superficial radiation therapy. Eliminating reimbursement for imaging would make it financially unfeasible for many dermatologists to provide image-guided treatment.
Let’s be clear about what that means.
It means arguing that seeing the cancer beneath the surface while targeting it provides no meaningful value.
In an era where precision defines modern medicine, some are advocating for less visibility, not more.
At the same time, ASTRO, representing radiation oncologists and the hospital systems where they practice, has lobbied to require additional certification for dermatologists who treat skin cancer with radiation therapy. Dermatologists were the first physicians to use radiation to treat cancer. They have been safely treating skin cancer with radiation for decades. Over 140,000 patients have already received image-guided SRT without the proposed new barrier.
If implemented, these restrictions would not eliminate radiation therapy. They would shift it.
Patients would be redirected to hospital-based settings where electron beam treatments are typically delivered at significantly higher cost, often multiple times the cost of office-based IGSRT, with greater logistical burden and, in many cases, less favorable cosmetic outcomes.
When professional societies representing procedural specialists and hospital-based physicians advocate policies that restrict lower-cost, office-based alternatives, it is reasonable to ask: who benefits?
This is no longer a debate about science alone. It is a debate about economics, control, and access.
No one disputes that Mohs surgery is an excellent treatment option for many patients. No one disputes that radiation oncology plays a critical role in cancer care. The issue is whether emerging, evidence-supported technology should be constrained not because it fails patients, but because it disrupts revenue streams.
History teaches us that when financial incentives collide with innovation, adoption slows.
And when adoption slows down, patients wait.
The AMA and CMS now face a defining decision. They can side with entrenched interests seeking to limit competition, or they can prioritize patient access to clinically validated, cost-effective, office-based innovation.
If we fail to learn from the past, the pattern will be repeated.
Evidence will exist.
Barriers will rise.
Patients will pay.
Dermatology does not need to become the next cautionary tale in medical history.
This is the moment to break the cycle.